The South African Health Products Regulatory Authority (SAHPRA), in partnership with Bayer, is overseeing the recall of a Yaz Plus contraceptive batch distributed last year due to an incorrect pill arrangement.
In a statement, Dr Boitumelo Semete-Makokotlela, CEO of SAHPRA, said they are aware of the quality issue regarding Batch No. WEW96J, that was distributed on November 9 and 24 2023, has an incorrect arrangement of tablets within some packs, thus affecting the efficacy of the affected batch.
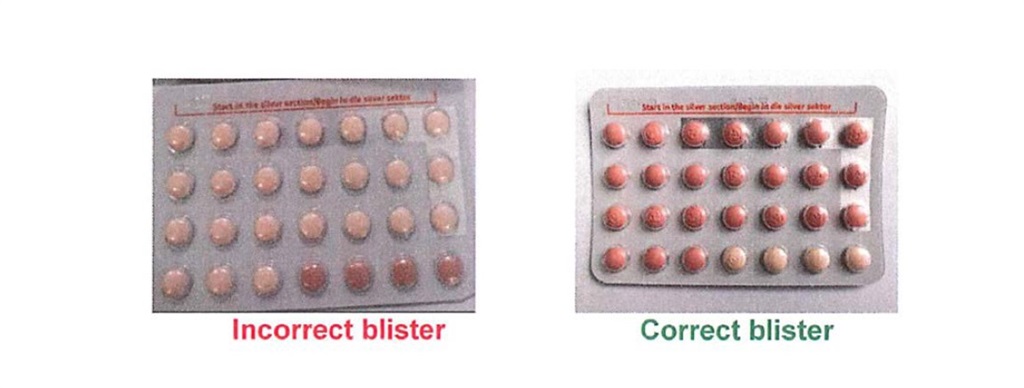
Semete-Makokotlela added, “Instead of containing 24 pink film-coated hormone tablets and 4 light orange hormone-free tablets, the affected packs contain 24 light orange hormone-free tablets and 4 pink film-coated hormone tablets.”
She further stated that the recall is a necessary action to protect the public from the possible inefficacy of the affected batch.
“We urge members of the public who are in possession of YAZ PLUS Batch No. WEW96J to discontinue use and to return the packaging to their pharmacists for a replacement with a pack from an unaffected batch or a refund,” urged Semete-Makokotlela.
“Members of the public who are using this contraceptive are urged to check the batch number of their current tablet packs, stop consuming the tablet, return affected packs to their pharmacist, and consult with their healthcare professional.”