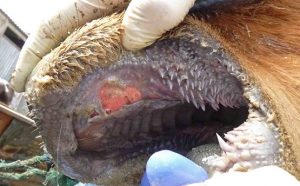
Agriculture minister John Steenhuisen has welcomed a Gauteng High Court decision that affirmed the department’s authority to regulate and manage South Africa’s response to foot-and-mouth disease (FMD).
The court on Tuesday did not grant urgent relief sought by business organisation Sakeliga and others, who had applied for permission to procure and administer FMD vaccines outside the established national framework.
Instead, the matter has been postponed to allow the department to finalise its vaccination scheme within a defined timeframe.
ALSO READ: One million FMD vaccines arrive in SA amid national outbreak
Steenhuisen said the outcome reinforced the principle that South Africa’s biosecurity response must be coordinated, science-based, and aligned with national and international standards.
“This matter was never simply about access to vaccines. It was about ensuring that South Africa’s response to FMD remains credible, coordinated, and compliant with the legal framework that protects both our national herd and our export markets,” he said in a statement.
The minister said the attempt to secure urgent court intervention to bypass established regulatory processes was both premature and misdirected.

He said the court recognised there were no grounds for urgent intervention that would disrupt the state’s ongoing disease control strategy.
The department has been working on developing a formal vaccination scheme, which has gone through consultation with the ministerial task team and industry stakeholders.
“The scheme will provide a structured mechanism for broader participation, while maintaining the necessary safeguards around traceability, vaccine integrity, and disease control,” Steenhuisen said.
He said vaccine procurement, distribution and administration was continuing under strict regulatory oversight, with additional consignments of vaccines already secured.
The minister said the department remained committed to working with industry stakeholders within the framework of the law, but would not compromise on the principles that underpin an effective biosecurity system.





You must be logged in to post a comment.